Authors:
Yujiro Fukuda1,2, Hidenori Suzuki2, Nobuhiro Hanai2, Hitoshi Hirakawa2, Taijiro Ozawa2, Eiichi Sasaki3, Yasushi Yatabe3, Hiroshi Yamashita1 and Yasuhisa Hasegawa2*
1Department of Otolaryngology, Yamaguchi University Graduate School of Medicine, 1-1-1, Minamikogushi, 755-8505 Ube, Japan
2Department of Head and Neck Surgery, Aichi Cancer Center Hospital, 1-1, Kanokoden, Chikusa-Ku, 464-8681 Nagoya, Japan
3Department of Pathology and Molecular Diagnostics, Aichi Cancer Center Hospital, 1-1, Kanokoden, Chikusa-Ku, 464-8681 Nagoya, Japan
Received: 30 September, 2016; Accepted: 12 October, 2016; Published: 13 October, 2016
Yasuhisa Hasegawa, Aichi Cancer Center Hospital, 1-1, Kanokoden, Chikusa-Ku, 464-8681 Nagoya, Japan, Tel: +81 52 762 6111; (x3104); Fax: +81-52-764-2944; E-mail:
Fukuda Y, Suzuki H, Hanai N, Hirakawa H, Ozawa T, et al. (2016) Prediction of Positive Surgical Margins in Sinonasal Tract Squamous Cell Carcinoma. Arch Otolaryngol Rhinol 2(1): 056-060. DOI: 10.17352/2455-1759.000026
© 2015 Fukuda Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Sinonasal tract; Squamous cell carcinoma; Positive surgical margin; Clinical N stage; Total maxillectomy
Background: In patients with clinical T4 (cT4) squamous cell carcinoma (SCC) of sinonasal tract who received surgery, the relationship between positive surgical margins and positive clinical N stage as diagnosed by the presence of cervical lymph node metastasis has not been investigated so far. Therefore, we investigated the relationship between positive surgical margins and preoperative parameters in patients with cT4 SCC of the sinonasal tract following surgery.
Methods: Forty-one patients who underwent surgery for cT4 SCC of the sinonasal tract were investigated and survival rates were calculated using the Kaplan-Meier method. The relationship between surgical margins and preoperative parameters was analyzed.
Results: Both positive surgical margins and clinical N stage were significantly correlated with shorter survival rate by log-rank test.
Conclusion: Positive surgical margins can be predicted based on clinical N stage in patients with cT4 SCC of the sinonasal tract.
Introduction
Squamous cell carcinoma (SCC) of the sinonasal tract with clinical T4 (cT4) classification, including paranasal sinuses and nasal cavity, usually has a worse prognosis than that with clinical T1 to T3. The 5-year overall survival (OS) rate of patients with cT4 SCC of the sinonasal tract who receive radical therapies, such as surgery and chemoradiotherapy, generally ranges from 32.0% to 75.0% [1-7]. However, a number of investigators has attempted to develop an accurate prognosis of SCC of the sinonasal tract using several approaches, such as clinical and pathological parameters [1-5].
Positive clinical N stage diagnosed based on the presence of cervical lymph node metastasis from clinical findings pre-surgery for SCC of the sinonasal tract leads to reduced rates of OS, and the rate of patients with SCC of the sinonasal tract and positive clinical N stage ranges from 3.3% to 36.7% [8-11]. Similarly, having positive surgical margins diagnosed by pathological findings following surgery for SCC of the sinonasal tract also leads to reduced rates of OS [2,5,12-16] and the rate of positive surgical margins in patients with SCC of the sinonasal tract ranges from 10.5% to 63.5% [14-17]. Of note, among patients with SCC of the sinonasal tract, the 5-year OS rate among those with positive surgical margins (0% - 32.8%) was significantly shorter than among those with negative surgical margins (65.7% - 81.8%) [13,15,18].
The relationship between positive surgical margins and preoperative parameters before surgery has been investigated in several cancers other than SCC of the sinonasal tract [19,20]. Age can predict positive surgical margins patients in cutaneous melanoma of the head and neck, and the rate of positive clinical N stage among patients with positive surgical margins is greater than that among those with negative surgical margins in subjects with SCC of the oral cavity [19,20]. To our knowledge, however, the relationship between positive surgical margins and positive clinical N stage in patients with cT4 SCC of the sinonasal tract following surgery has not been investigated.
Here, we investigated the relationship between positive surgical margins and preoperative parameters in patients with cT4 SCC of the sinonasal tract following surgery and determined whether or not positive clinical N stage can predict positive surgical margins using univariate and multivariate analyses with adjustments for clinical parameters.
Materials and Methods
Patients and treatments
Between January 2001 and December 2011, 52 patients underwent surgery for malignant neoplasms of the sinonasal tract, including SCC and non-SCC with cT4, at the Department of Head and Neck Surgery in Aichi Cancer Center Hospital. We excluded 11 patients who did not have pathologically diagnosed SCC, resulting in enrollment of 41 patients with pathologically diagnosed cT4 SCC of the sinonasal tract. All patients gave informed consent for each examination and treatment.
Routine clinical examinations and blood chemistry were performed on the first visit to our institution. Clinical T and N classifications were diagnosed by physical examination, nasopharyngoscopy, and enhanced cervical computed tomography (CT) or magnetic resonance imaging (MRI). Positive clinical N stage was defined as findings on enhanced cervical CT of ringed enhancement or short distance of lymph node ≥ 10 mm. As no patients showed evidence of either distant metastasis or second primary cancers on chest CT, we performed positron emission tomography (PET) or PET/CT when possible. Diagnoses were made according to the clinical TNM classification of the Union for International Cancer Control (6th edition) [21].
Unresectable disease was defined by the presence of distant metastasis or the involvement of the sphenoid sinus, clivus, cavernous sinus, both orbits, and internal carotid artery. Resectable disease was defined as a tumor that could be resected in an en bloc fashion [22]. All patients underwent both en bloc resection of primary tumors and free-flap reconstructive procedures, as previously described [22]. Total maxillectomy was conducted for 11 patients and extended total maxillectomy for 30 (5 with orbital exenteration and 25 with skull base resection). Thirty-seven of the 41 patients received induction chemotherapy (ICT), with the majority of regimens consisting of 5-fluorouracil (800mg/m2/day, day1-5) and cisplatin (80mg/m2/day, day6), before surgery for tumor shrinkage and distant metastasis suppression. ICT has been received to nearly all patients. However, some of the patients did not receive the ICT for its rejection or other reasons.
Thirty-two (78.1%) of the 41 patients were clinically diagnosed with no lymph node metastasis before surgery, and 10 of the 41 patients underwent prophylactic neck dissection while 9 (21.9%) who were N-positive underwent neck dissection. Prophylactic neck dissection was performed at the discretion of the attending physician.
Resected specimens consisted of 5-mm thick sections cut from the tumor by a head and neck surgeon that were then fixed with 10% formalin for several days. After macroscopic evaluation of surgical margins by both an experienced pathologist and head and neck surgeon, sections were stained with hematoxyline and eosine (H&E). Pathological diagnoses were made by two experienced pathologists who then compiled all reports. Surgical margins were defined as follows [20]. free margin, tumor at least 5 mm from the surgical margin; closed margin, tumor less than 5mm from the surgical margin; and involved margin, tumor present in the surgical margin. In this study, involved margins were categorized as positive surgical margins, and both free and closed margins as negative surgical margins.
Thirty-four (82.9%) of the 41 patients had negative surgical margins while 7 (17.1%) had positive surgical margins. When possible, we planned postoperative radiation therapy (PORT) at a total dose of 60 Gy with or without chemotherapy for patients with positive surgical margins. However, 24 patients did not receive PORT due to prolonged wound healing after surgery or lack of patients consent. Clinical characteristics of patients are shown in Table 1. Preoperative enhanced CT images and H&E stains of one randomly selected patient with positive surgical margins and one with negative surgical margins are shown in Figure 1.
-
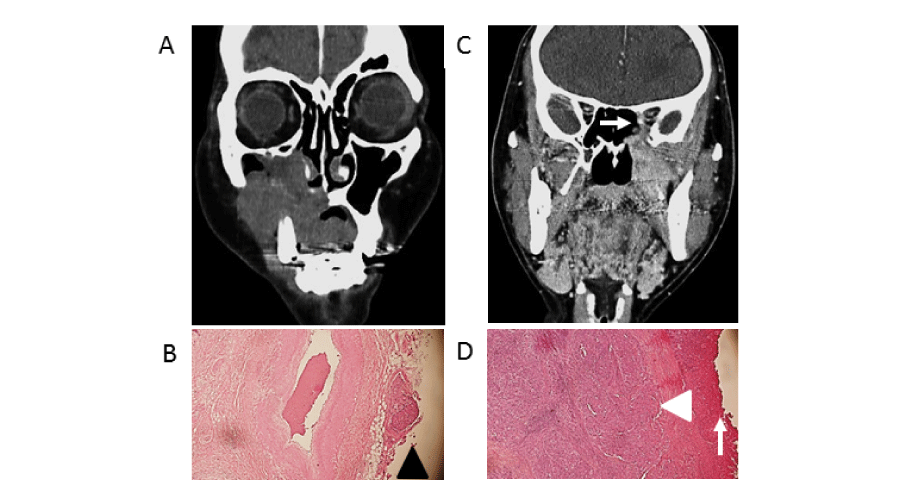
Figure 1:
(A, C) Preoperative enhanced computed tomography images (coronal section), (B, D) hematoxylin-eosin stain of surgically resected tissues. Original magnification, x10. (A, B) Case of clinical T4aN2cM0 that received extended total maxillectomy with skull base resection. (C, D) Case of cT4bN0M0 with invasion of orbital apex (white arrow) that received extended total maxillectomy with orbital exenteration. (B) Surgical margin positivity was pathologically diagnosed based on the presence of cancer cells at the border (black arrowhead). (D) Negative surgical margins were pathologically diagnosed based on the absence of cancer cells at the border (white arrow), and cancer cells were covered by surrounding normal cells (white head).
After treatment completion, patients were followed up at our outpatient clinic. Effort was made to identify patients with early locoregional recurrence and perform radical salvage therapy.
Statistical analysis
Statistical analysis was performed using JMP 9 software (SAS Institute Inc, Cary, NC, USA). The Kaplan-Meier method was used to estimate overall survival (OS), disease-free survival (DFS), local recurrence-free survival (LRFS), regional recurrence-free survival (RRFS), distant metastasis-free survival (DMFS), and locoregional recurrence-free survival (LRRFS) curves. We defined survival time as the period from surgery to target events (see below), which were death for OS, date of recurrence at any sites or distant metastasis for DFS, date of local recurrence for LRFS, date of regional recurrence for RRFS, date of distant metastasis for DMFS, and date of local or regional recurrence for LRRFS.
In univariate survival analysis of all patients, two groups separated based on clinical and pathological characteristics (clinical T and N classification, type of surgery, surgical margin, ICT) were assessed using the log-rank test.
In univariate analysis by chi-square test, the relationship between patients with positive surgical margins and those with negative surgical margins based on preoperative parameters (clinical T and N classification, type of surgery, ICT) was analyzed. In multivariate analysis involving logistic regression, the relationship between patients with positive surgical margins and those with negative surgical margin based on preoperative parameters (clinical T classification, clinical N classification, type of surgery and ICT) was analyzed. P value less than 0.05 was considered significant.
Results
Clinical course
The median follow-up period for all patients was 52 months (range: 1-153 months). Thirteen (31.7%) of the 41 patients died by the end of this study. Eleven of those 13 (26.8% vs. all) died of SCC of the sinonasal tract, while 2 patients (4.9% vs. all) died of pneumonia and respiratory dysfunction. Ten patients with negative clinical N stage were recieved prophylactic neck dissection, and all of patients were pathologically negative N stage. Nine patients with positive clinical N stage were recieved neck dissection, and 5 patients were pathologically positive N stage. Thirteen (31.7%) of the 41 patients had recurrence or distant metastasis. Sites of recurrence or distant metastasis were as follows: local alone, 5 patients (12.2% vs. all); regional site alone, 2 patients (4.9% vs. all); distant site alone, 2 patients (4.9% vs. all); local and regional sites, 1 patient (2.4% vs. all); local and distant sites, 1 patient (2.4% vs. all); regional and distant sites, 1 patient (2.4% vs. all); and local, regional, and distant sites, 1 patients (2.4% vs. all). Five of the patients with either recurrence or distant metastasis (e.g. solitary lung metastasis) underwent salvage surgery.
Survival analyses
The 5-year survival rates for all patients were as follows: OS (67.5%), DFS (66.0%), LRFS (78.9%), RRFS (86.1%), LRRFS (71.2%) and DMFS (82.9%). In univariate survival analysis of all patients, those with positive surgical margins (n=7) were significantly correlated with shorter OS (p <.01), DFS (p <.01), LRFS (p <.01), RRFS (p <.01), LRRFS (p <.01) and DMFS (p <.01) than those with negative surgical margins (n=34), and those with positive clinical N stage (n=9) was significantly correlated with shorter RRFS (p <.03) and LRRFS (p <.04) than those with negative clinical N stage (n=32). Results of these univariate survival analyses are shown in Figure 2.
-
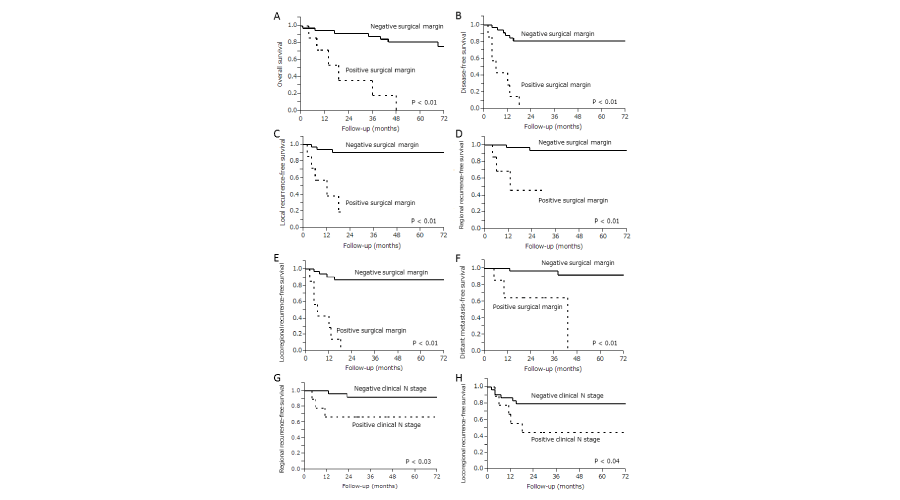
Figure 2:
Kaplan-Meier curves in 41 patients with clinical T4 classification squamous cell carcinoma of sinonasal tract. Significant correlation was noted between the positive surgical margin group and shorter (A) overall survival, (B) disease-free survival, (C) local recurrence-free survival, (D) regional recurrence-free survival, (E) locoregional recurrence-free survival, and (F) Distant metastasis-free survival than that of negative surgical margin group. Being in the positive clinical N stage group significantly correlated with shorter (G) regional recurrence-free survival and (H) locoregional recurrence-free survival. Log-rank test used for statistical analysis.
We performed multivariate analysis to examine whether or not positive surgical margins were independent of positive clinical N stage for OS, DFS, LRFS, RRFS, LRRFS and DMFS. Patients with positive surgical margins exhibited a significant correlation for OS (p <.01), DFS (p <.01), LRFS (p <.01), RRFS (p <.05) and LRRFS (p <.01). Three- and five-year OS rates among patients with positive surgical margins were 17.9% and 0%, respectively, while those among patients with negative surgical margins were 87.5% and 80.5%. The results of multivariate survival analysis are shown in Table 2.
-

Table 2:
Multivariate analysis* of patient survival.
Chi-square test and logistic regression analysis
The relationship between patients with positive and negative surgical margins based on preoperative parameters in both univariate and multivariate analyses is shown in Table 3. In univariate analysis, positive clinical N stage was significantly associated with positive surgical margins by chi-square test (p <.02). In multivariate analysis of logistic regression analysis with adjustments for preoperative parameters, positive clinical N stage was significantly associated with positive surgical margins (hazard ratio: 7.92, 95% confidence interval: 1.17-73.6, p <.04).
-

Table 3:
Relationship between patients with positive and negative surgical margins based on preoperative parameters in univariate* and multivariate† analyses.
Discussion
To our knowledge, we have shown for the first time that positive clinical N stage is significantly associated with positive surgical margins in patients with cT4 SCC of the sinonasal tract following surgery.
Patients with positive clinical N stage were more significantly correlated with shorter RRFS and LRRFS than those with negative clinical N stage. Both clinical T and N classifications in various types of cancer, including sinonasal tract SCC, are widely accepted as prognostic parameters and patients with positive clinical N positive were more significantly correlated with shorter survival, such as OS, than those with negative clinical N stage [5,7,9,15,23,24]. For example, Homma et al. [9], reported that in 54 patients receiving intra-arterial cisplatin infusion and concomitant radiotherapy for maxillary sinus cancer (cT4 account for 72.2%), those with positive clinical N stage were more significantly correlated with shorter OS than those with negative clinical N stage. In 4994 sinonasal SCC patients who received radical therapy, those with positive clinical N stage were associated with lower OS rate [24]. Our result of the relationship between positive clinical N stage and shorter survival is consistent with these reports [5,7,9,15, 3,24].
In the present study, groups of patients with positive surgical margins were more significantly correlated with shorter OS than that those with negative surgical margins. In many reports of cancer of the sinonasal tract, including SCC and non-SCC treated by radical surgical resection, patients with positive surgical margins were more significantly correlated with shorter OS than those with negative surgical margins [5,13,15,16]. For example, Thomas et al. [13], reported that in 121 patients who received surgery for cT4 SCC of the maxilla and midface, that those with positive surgical margins were more significantly correlated with shorter OS than those with negative surgical margins. In 1307 malignant skull base tumor patients who received surgery, those with positive surgical margins were significantly correlated with shorter OS than those with negative surgical margins [15]. Our result demonstrating the association between shorter OS and positive surgical margins is consistent with these studies [5,13,15,16].
In the present study, respective 5-year OS rates among patients with positive surgical margins and negative surgical margins were 0% and 80.5%. In previous reports of SCC of the sinonasal tract, including SCC and non-SCC treated by radical surgical resection, respective 5-year OS rates among patients with positive and negative surgical margins ranged from 0% to 32.8% and 65.7% to 81.8% [13,15,16,18]. In 121 patients who received surgery for cT4 SCC of the maxilla and midface, 5-year OS rates among patients with positive or negative surgical margins were 0% and more than 60%, respectively [15]. Our findings for 5-year OS of patients with positive and negative surgical margins are therefore consistent with those of previous studies [13,15,16,18].
Several groups researching oral and oropharyngeal cancer have demonstrated that patients with positive surgical margins are strongly associated with both pathological T and N classification [20,25]. Two groups studying cutaneous melanoma of the head and neck and SCC of the oral cavity recently demonstrated a relationship between positive surgical margins and preoperative parameters [19,20]. Christophel et al. [19], reported in patients with cutaneous melanoma of head and neck that age predicts positive surgical margins, and Girardi et al. [20], described in patients with SCC of the oral cavity that the rate of positive N stage in the positive surgical margin group was greater than that in the negative surgical margin group.
To our knowledge, among patients with cT4 SCC of the sinonasal tract following surgery, the relationship between positive surgical margin and clinical N stage has not been investigated. We hypothesized that positive clinical N stage before surgery in patients with cT4 SCC of sinonasal tract is related to positive surgical margins, as both positive clinical N stage and surgical margin influence shorter rates of survival such as OS [5,7,13,15,16,24]. In our present study, positive clinical N stage was significantly correlated with positive surgical margins in both univariate and multivariate analyses. This finding suggests to us being kept in mind that tumors with lymph node metastasis are locally more aggressive at surgery. In addition, this finding suggests that positive clinical N stage before surgery may be an effective parameter for identifying patients at high risk of positive surgical margins. For example, it may be suggest a beneficial impact of ICT and/or PORT on these high risk patients. However, limitations to our present study that warrant attention include its retrospective study design and relatively small number of subjects. Analysis of a larger number of patients is therefore required in future studies.
Conclusion
We have shown in patients with cT4 SCC of the sinonasal tract who received surgery that positive clinical N stage was significantly associated with positive surgical margins in univariate and multivariate analyses. Preoperatively, positive clinical N stage might prove useful in identifying patients with cT4 SCC of the sinonasal tract at high risk of positive surgical margins.
Acknowledgements
This study was partially supported by a Health and Labour Sciences Research Grant for Clinical Cancer Research from the Ministry of Health, Labour and Welfare, Japan.
-

Figure 1:
27 year-old man with gradual onset of left -sided facial weakness five days after accidental blunt head trauma. High-resolution TB coronal CT scan at the level of the labyrinthine segment of the FN: no bone fracture.
-
-
- Mendenhall WM, Amdur RJ, Morris CG, Kirwan J, Malyapa RS, et al. (2009) Carcinoma of the nasal cavity and paranasal sinuses. Laryngoscope 119: 899-906 .
- Mine S, Saeki N, Horiguchi K, Hanazawa T, Okamoto Y (2011) Craniofacial Resection for Sinonasal Malignant Tumors: Statistical Analysis of Surgical Outcome over 17 Years at a Single Institution. Skull Base 21: 243-248 .
- Dirix P, Nuyts S, Geussens Y, Jorissen M, Vander Poorten V, et al. (2007) Malignancies of the nasal cavity and paranasal sinuses: long-term outcome with conventional or three-dimensional conformal radiotherapy. Int J Radiat Oncol Biol Phys 69: 1042-1050 .
- Guntinas-Lichius O, Kreppel MP, Stuetzer H, Semrau R, Eckel HE, et al. (2007) Single modality and multimodality treatment of nasal and paranasal sinuses cancer: a single institution experience of 229 patients. Eur J Surg Oncol 33: 222-228 .
- Mirghani H, Mortuaire G, Armas GL, Hartl D, Aupérin A, et al. (2013) Sinonasal cancer: Analysis of oncological failures in 156 consecutive cases. Head Neck 36: 667-674 .
- Dalal AJ, McLennan AS (2013) Cervical metastases from maxillary squamous cell carcinoma: retrospective analysis and of the literature. Br J Oral Maxillofac Surg 51: 702-706 .
- Kano S, Hayashi R, Homma A, Matsuura K, Kato K, et al. (2013) Effect of local extension sites on survival in locally advanced maxillary sinus cancer. Head Neck 36: 1567-1572.
- Cantù G, Bimbi G, Miceli R, Mariani L, Colombo S, et al. (2008) Lymph node metastases in malignant tumors of the paranasal sinuses: prognostic value and treatment. Arch Otolaryngol Head Neck Surg 134: 170-177 .
- Homma A, Sakashita T, Yoshida D, Onimaru R, Tsuchiya K, et al. (2013) Superselective intra-arterial cisplatin infusion and concomitant radiotherapy for maxillary sinus cancer. Br J Cancer 109: 2980-2986 .
- Rinaldo A, Ferlito A, Shaha AR, Wei WI (2002) Is elective neck treatment indicated in patients with squamous cell carcinoma of the maxillary sinus? Acta Otolaryngol 122: 443-447 .
- Kruse AL, Grätz KW (2009) Cervical metastases of squamous cell carcinoma of the maxilla: a retrospective study of 9 years. Head Neck Oncol 1: 28 .
- Eldeeb H, Macmillan C, Elwell C, Hammod A. (2012) The effect of the surgical margins on the outcome of patients with head and neck squamous cell carcinoma: single institution experience. Cancer Biol Med 9: 29-33 .
- Mücke T, Loeffelbein DJ, Hohlweg-Majert B, Kesting MR, Wolff KD, et al. (2009) Reconstruction of the maxilla and midface-surgical management, outcome, and prognostic factors. Oral Oncol 45: 1073-1078 .
- Bobinskas AM, Wiesenfeld D, Chandu A (2014) Influence of the site of origin on the outcome of squamous cell carcinoma of the maxilla-oral versus sinus. Int J Oral Maxillofac Surg 43: 137-141 .
- Patel SG, Singh B, Polluri A, Bridger PG, Cantu G, et al. (2003) Craniofacial surgery for malignant skull base tumors: report of an international collaborative study. Cancer 98: 1179-1187 .
- Bristol IJ, Ahamad A, Garden AS, Morrison WH, Hanna EY, et al. (2007) Postoperative radiotherapy for maxillary sinus cancer: long-term outcomes and toxicities of treatment. Int J Radiat Oncol Biol Phys 68: 719-730 .
- Janjua OS, Ahmed W, Qureshi SM, Khan TS, Ahmed A, et al. (2013) Assessment of margins in resection specimens for head and neck malignancies. J Coll Physicians Surg Pak 23: 265-268 .
- Yang Z, Deng R, Sun G, Huang X, Tang E (2013) Cervical metastases from squamous cell carcinoma of hard palate and maxillary alveolus: A retrospective study of 10 years. Head Neck 36: 969-975.
- Christophel JJ, Johnson AK, McMurry TL, Park SS, Levine PA. (2013) Predicting positive margins in resection of cutaneous melanoma of the head and neck. Laryngoscope 123: 683-688 .
- Girardi FM, Zanella VG, Kroef RG. (2013) Correlation between clinical and pathological data and surgical margins in patients with squamous cell carcinoma of the oral cavity. Braz J Otorhinolaryngol 79: 190-195 .
- Sobin LH, Wittekind Ch. (2002) International Union Against Cancer (UICC): TNM classification of malignant tumors, 6th edition. Wiley-Liss, New York .
- Okada T, Saito K, Takahashi M, Hasegawa Y, Fujimoto Y, et al. (2008) En bloc petrosectomy for malignant tumors involving the external auditory canal and middle ear: surgical methods and long-term outcome. J Neurosurg 108: 97-104 .
- Sagheb K, Sagheb K, Taylor KJ, Al-Nawas B, Walter C (2013) Cervical metastases of squamous cell carcinoma of the maxilla: a retrospective study of 25 years. Clin Oral Investig 18: 1221-1227.
- Sanghvi S, Khan MN, Patel NR, Yeldandi S, Baredes S, et al. (2014) Epidemiology of sinonasal squamous cell carcinoma: a comprehensive analysis of 4994 patients. Laryngoscope 124: 76-83 .
- Sutton DN, Brown JS, Rogers SN, Vaughan ED, Woolgar JA (2003) The prognostic implications of the surgical margin in oral squamous cell carcinoma. Int J Oral Maxillofac Surg 32: 30-34 .









Table 1:
Clinical characteristics of patients.