Open Journal of Environmental Biology
Mosquito borne diseases constitute a major public health problem in the list of communicable diseases,
Adesina JM1*, Jose AR2 and Mobolade-Adesina TE2
1Environmental Biology Programme, Department of Science Laboratory Technology, Rufus Giwa Polytechnic, P. M. B. 1019, Owo, Ondo State, Nigeria
Cite this as
Adesina JM, Jose AR, Mobolade-Adesina TE (2019) Mosquitocidal potential of Chysophylum africanum seed extracts against human vector mosquitoes Anopheles stephensi (Liston) (Diptera: Culicidae). Open J Environ Biol 4(1): 019-022. DOI: 10.17352/ojeb.000012Mosquito borne diseases constitute a major public health problem in the list of communicable diseases, An. stephensi (Liston) is the primary vector of malaria in developing countries and improved methods of control are urgently needed. Larvicidal activity of Chysophylum africanum seed extracts was evaluated against An. stephensi. Fourth instar larvae were exposed to 100, 150 and 200µl concentrations of ethyl acetate, acetone and methanol extracts of C. africanum replicated thrice. Larval mortality was observed at 24 and 48 h after exposure. Results obtained show that the various extracts caused significant mortality effect with increased concentration and duration of exposure, with ethyl acetate extract invoking the maximum mortality (90%) within 24 hours of exposure at all concentrations tested, while acetone recorded the lowest mortality (33.85%) at 200µl. These findings indicate that seeds of C. africanum have larvicidal activity that can be exploited for the management of malaria vector. More studies are needed to determine phytochemical constituents and its mode of action for possible development of larvicides that are safe to use and environment friendly.
Introduction
Vector-borne diseases are one of the paramount contributors to human death and ill health in the tropics and subtropics. Every year, more than one billion people are infected and more than one million people die from vector-borne diseases including malaria, dengue, yellow fever and lymphatic filariasis [1].
Mosquitoes disseminate more sicknesses than any other group of arthropods and present an immense threat to millions of people, since they act as vectors for important parasites and pathogens, causing millions of death annually across the world [2-3].
Vector control remains the most effective measure and is often the only way to avert disease transmission and outbreaks because there are no vaccines for many vector-borne diseases and drug resistance is an increasing threat. One of the approaches to manage these diseases is to control the vectors for bringing about interruption in disease transmission. The control of mosquitoes at larval stage is considered as an efficient way in integrated vector management [4]. Therefore, the ideal method for controlling mosquito influx and its attendant diseases is the prevention of mosquito breeding by using appropriate larvicides or application of insecticides to larval habitats, destroying unwanted containers, and educating the public [5-7]. Larval stages of the mosquitoes are attractive targets for pesticides to control mosquito populations [8]. Hence, the advantages of targeting the larval stages are that mosquitoes are killed before they disperse to human habitations and that larvae, unlike adults, cannot change their behaviour to avoid control activities and also to moderate overall pesticide use in control of adult mosquitoes [9-10].
Synthetic insecticides are the first line of action owing to their quick action, but their incessant and indiscriminate use led to the resistance development and lasting residual effect on the environment which can be injurious to animals including human and higher rate of biological magnification [11-13].
Hostile environmental effects and the need to maintain a sustainable environment have created the need for environmental-safe, degradable and target specific-insecticides which remain the top research issue for scientists associated with alternative vector control [14]. The pursuit has been directed extensively to the plant kingdom. Many plant compounds have larvicidal, pupicidal and adulticidal activities, most being repellents, ovipositional deterrents and anti-feedants against both agricultural and medically important insect species [15]. In fact, many researchers have reported the efficacy of plant extracts or essential oils against mosquito larvae [16-17]. In view of the recently increased interest in developing plant origin insecticides as an alternative to chemical insecticides. The present study was carried out to determine the larvicidal activity of Chysophylum africanum seed extracts against malaria vectors An. stephensi.
Materials and Methods
Experimental location
The experiment was carried out in the Environmental Biology Laboratory, Department of Science Laboratory Technology, Rufus Giwa Polytechnic, Owo, Ondo State, Nigeria under ambient laboratory conditions of 30 ± 2°C temperature, 75 + 5% relative humidity and 12L:12D photoperiod.
Collection of mosquito larvae
The mosquito larvae were collected from stagnant water samples from drainage canals and temporary water pools around student hostels in the polytechnic using sterile wide mouth container. The identified An. stephensi mosquito larvae were kept in plastic bowl containing rain water, sprinkled with powder yeast to serve as food. The culture was maintained and reared in laboratory.
Plants collection and extraction
Seeds of C. africanum were collected from different locations in Owo metropolis and air dried for 4 weeks. Thereafter, the seeds were cracked to remove their endocarps which were further air dried for 5 days. The dried seeds were powdered mechanically using commercial electrical stainless steel blender and extracted sequentially with ethyl acetate, acetone and methanol in a soxhlet apparatus separately. The extracts were concentrated under reduced pressure of 22-26mm hg at 45º C using rotary evaporator in order to remove the extracting solvents [18].
Larvicidal activity of C. africanum extracts
The larvicidal activity of C. africanum extract was assessed by using the standard method as prescribed by World Health Organization [19]. From the stock solution, four different test concentrations (100, 150 and 200 µl with the control) were prepared and they were tested against third instar larvae of, An. stephensi based on its morphological features. Five larvae were introduced in 250ml plastic dishes cups containing 100 ml of rainwater and the required amount of plant extract was added. Controls were run simultaneously.
The larval mortality was observed and recorded after 24 and 48 h of post treatment. Larval percentage mortality was corrected using Abott formula [20]. For each experiment, three replicates were maintained at a time. The average mortality data were subjected to probit analysis for calculating LD90 [21].
Statistical analysis
Data from all replicates were pooled for analysis. LC90 values were calculated using SPSS software by probit analysis [22]. ANOVA was performed to determine the difference in larval mortality between concentrations. Results with P<0.05 level were considered to be statistically significant.
Results
The seed extracts of C. africanum exhibited significant larvicidal activity in all the tested concentrations against An. stephensi. Ethyl acetate extract evoked 90% larval mortality at all concentrations within 24 h of treatment, acetone and methanol extracts exhibited concentration and time of exposure dependent with the maximum mortality 33.85% and 51.93% recorded at 200µl respectively. Similar trend was observed 48 h after treatment (Table 1).
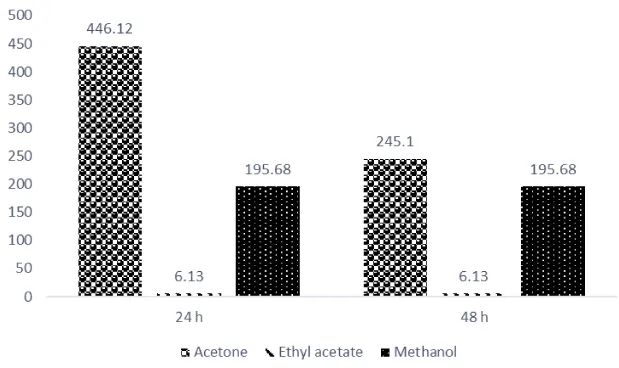
Lethal dose of the extracts was significantly influenced by the solvent used for the extraction. Results in figure 1 show that LD50 was maximum with ethyl acetate extract at both 24 and 48 h exposure time (6.13), distantly followed by methanol extract (195.58).
Discussion
Mosquitoes are the oldest human enemy and controlling them is of prime importance in recent years because of the numerous diseases caused by them. Around the world, the medical and economic burden caused by vector-borne diseases continue to grow as current control measures fail. The results of the present study may contribute to a decline in the application of synthetic insecticides, which in turn increase the opportunity for natural control of various medically important pests by botanical pesticides.
Results from the study clearly indicate that various C. africanum extracts significantly caused larvae mortality. This allude to the presence of phytochemicals, toxic to the survival of the larvae. The mortality recorded is directly proportional to increase in concentration level and period of exposure; also the larval mortality varies in relation to the extract used.
Extracts of the plants derived from specific solvents can influence the bioactivity, probably because of the active components present in large quantities [23]. It has been demonstrated that extraction of active biochemicals from plants confides upon the polarity of the solvents utilized. Polar solvent will extract polar molecules and non-polar solvents extract non-polar molecules [24].
Ethyl acetate is a moderately polar solvent (polarity index of 4.4) that mainly extracts steroids, alkaloids, etc. Those biochemicals that were extracted using moderately polar solvents were seen to give good results as reported by a few bioassays. This corroborates with the findings of the present study. Thus, different solvent types can significantly affect the potency of extracted plant compounds and there is difference in the chemo-profile of the plant species [25].
Plant extracts might have some complex mixture of biocidal active compounds, which may jointly or independently contribute to the mortality and delayed growth of larvae [26]. Rey et al. [27] and David et al. [28] found that botanical derivatives primarily affect the midgut epithelium and secondarily affect the gastric caeca and the malpighian tubules in mosquito larvae.
Conclusion
The results of this study indicate that C. africanum ethyl acetate extract has a promising effect to control mosquito larvae and may be an effective alternative to conventional synthetic insecticides. The ethyl acetate extract of this plant may be fractioned in order to locate the particular bioactive toxic agent responsible for larval toxicity.
- WHO (2014) A global brief on vector-borne diseases. WHO/DCO/WHD. Link: http://bit.ly/2Gd3rzx
- Rueda LM (2008) Global diversity of mosquitoes (Insecta: Diptera: Culicidae) in freshwater. Hydrobiologia 595: 477-487. Link: http://bit.ly/2Xz17bR
- Benelli G (2015) Research in mosquito control: current challenges for a brighter future. Parasitol Res 114: 2801-2805. Link: http://bit.ly/2XwGGfz
- Rutledge CR, Clark F, Curtis A (2003) Larval mosquito control. Technical Bulletin of the Florida Mosquito Control Association 4: 16-19.
- World Health Organization (2005) Guidelines for laboratory and field testing of mosquito larvicides. Link: http://bit.ly/2LHVENs
- Tiwary M, Naik SN, Tewary DK, Mittal PK, Yadav S (2007) Chemical Composition and Larvicidal Activities of the Essential Oil of Zanthoxylum armatum DC (Rutaceae) Against three Mosquito Vectors. J Vector Borne Dis 44: 198-204. Link: http://bit.ly/2L76hKf
- Tennyson S, Ravindran KJ, Arivoli S (2012) Screening of twenty-five plant extracts for larvicidal activity against Culex quinquefasciatus Say (Diptera: Culicidae). Asian Pacific Journal of Tropical Biomedicine 2: S1130- S1134. Link: http://bit.ly/2JiRw4S
- Amer A, Mehlhorn H (2006) Larvicidal effects of various essential oils against Aedes, Anopheles and Culex larvae (Diptera, Culicidae). Parasitol Res 99: 466-472. Link: http://bit.ly/2YESPjR
- Killeen GF, Fillinger U, Knols BG (2002) Advantages of larval control for African malaria vectors: low mobility and behavioural responsiveness of immature mosquito stages allow high effective coverage. Malaria Journal 1:8. Link: http://bit.ly/32e1wnB
- Gleiser RM, Zygadlo JA (2007) Insecticidal properties of essential oils from Lippia turbinata and Lippia polystachya (Verbenaceae) against Culex quinquefasciatus (Diptera: Culicidae). Parasitology Research 101: 1349-1354. Link: http://bit.ly/2L8Lzd7
- Kumar S, Wahab N, Mishra M, Warikoo R (2012) Evaluation of 15 local plant species as larvicidal agents against an Indian strain of dengue fever mosquito, Aedes aegypti L. (Diptera: Culicidae). Front Physiol 3: 104. Link: http://bit.ly/2RZIY5Q
- Raghavendra K (2002) Chemical insecticides in malaria vector control in India. ICMR Bulletin. 32: 1-7. Link: http://bit.ly/32freYQ
- Borase HP, Patil CD, Salunkhe RB, Narkhede CP, Salunke BK, et al. (2013) Phyto-synthesized silver nanoparticles: A potent mosquito biolarvicidal agent. Journal of Nanomedicine and Biotherapeutic Discovery 3: 1-7. Link: http://bit.ly/2Xu7lhY
- Isman MB (2000) Plant essential oils for pest and disease management. Crop Protection 19: 603-608. Link: http://bit.ly/30dw4UG
- Babu R, Murugan K (1998) Interactive effect of neem seed kernel and neem gum extracts on the control of Culex quinquefasciatus say. Neem News Letters 15: 9-11.
- Ghosh A, Chowdhury N, Chandra G (2012) Plant extracts as potential larvicides. Indian Journal of Medical Research 135: 581-598. Link: http://bit.ly/2FYgVPa
- Sukumar K, Perich MJ, Boobar LR (1991) Botanical derivatives in mosquito control: A review. American Mosquito Control Association 7: 210-237. Link: http://bit.ly/2XwQ3Mq
- Vogel AL (1978) Text Book of Practical-Organic Chemistry. The English Language Book Society and Langman London. 1363.
- World Health Organization (2005) Guidelines for laboratory and field testing of mosquito larvicides. Communicable disease control, prevention and eradication, WHO pesticide evaluation scheme. WHO, Geneva, WHO/CDS/WHOPES/GCDPP/1.3. 2005.
- Abbott WS (1925) A Method of Computing the Effectiveness of an Insecticide. Journal of Economic Entomology 18: 265–267. Link: http://bit.ly/2Xu8tSK
- Finney DJ (1979) Probit analysis. Cambridge University Press, London. 68–72.
- SPSS (2007) SPSS for windows, Version 11.5. SPSS, Chicago, Illinois, USA.
- Oliveira PV, Ferreira JC, Moura FS, Lima GS, de Oliveira FM, et al. (2010) Larvicidal activity of 94 extracts from ten plant species of north eastern of Brazil against Aedes aegypti L. (Diptera: Culicidae). Parasitol Res 107: 403-407. Link: http://bit.ly/2LJekwb
- Annie SW, Raveen R, Paulraj MG, Samuel T, Arivoli S (2016) Screening of Hydrilla verticillata (L. F.) Royle (Hydrocharitaceae) crude leaf extracts for larvicidal efficacy against the filarial vector Culex quinquefasciatus say (Diptera: Culicidae). International Journal of Entomology Research 1: 43-48. Link: http://bit.ly/2JjasjT
- Ghosh A, Choudhary N, Chandra G (2012) Plant extracts as potential mosquito larvicides. Indian J Med Res 135: 581-598. Link: http://bit.ly/2FYgVPa
- Tiwari P, Kumar B, Kaur M, Kaur G, Kaur H (2011) Phytochemical screening and Extraction: A Review. Internationale Pharmaceutical Sciencia 1: 90-106. Link: http://bit.ly/2L9uD62
- Rey D, Cuany A, Pautou MP, Meyran JC (1999) Differential sensitivity of mosquito taxa to vegetable tannins. Journal of Chemical Ecology 25: 537-548. Link: http://bit.ly/2L7ldrR
- David JP, Rey D, Pauntou MP, Meyran JC (2000) Differential toxicity of leaf litter to dipteran larvae of mosquito developmental sites. Journal of Invertebrate Pathology 75: 9-18. Link: http://bit.ly/2YFeGI7

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Save to Mendeley
Save to Mendeley
